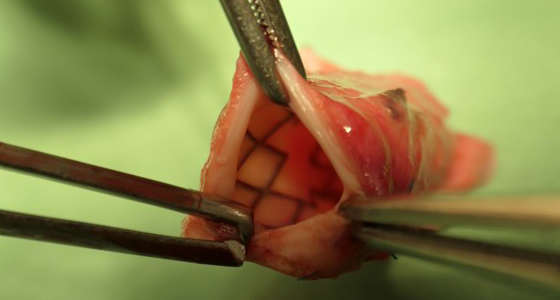
Micro-Drilling for new Stenosis-Treatment
With an innovative method of laser-drilling micro-holes in balloon catheters, MeKo now offers its customers the possibility to develop new and improved components for the treatment of stenosis (vascular constrictions). The special feature: MeKo is capable of producing perforated balloons with holes having incredibly small diameters of only 4-8 µm.
Stenosis can occur in arteries due to fat and calcium deposits within the blood vessel and is commonly treated with a stent. Recently, whenever possible and reasonable, balloons have been used more frequently to dilate the stenosis, rather than implanting a stent.
Drug-coated balloons (DCBs) deliver antiproliferative drugs to the vessel wall while dilating the stenosis to prevent cell proliferation during healing of the damaged vessel.
However, DCBs have several disadvantages: First of all, the drug adhering to the balloon has a very limited shelf life. In addition, the DCB loses some of its coating as it travels through the blood vessels to the stenosis. As a result, the final drug dose is unpredictable. Furthermore, the drug is predominantly released linearly from the folds of the balloon, resulting in an uneven drug delivery with some areas receiving more drugs than others.
To avoid these and other disadvantages of DCBs, MeKo has developed a new type of balloon catheter, micro-drilling several hundred micro-holes into the balloon called Drug Delivery Balloon (DDB). The advantages of the DDB are obvious: The balloons are only filled with drugs while being used. Until then, they have an almost unlimited shelf life and any drug won’t be released before the blood vessel is dilated.
Due to freely customizable and individual micro-hole patterns and MeKo’s highly precise manufacturing capabilities, the drug release will occur exactly as intended and uniformly throughout the entire balloon.
Very narrow manufacturing tolerances of 0.220 µm (0.0000086”) and quality control process inspecting every single hole allow a highly reproducible and therefore safe procedure.